Over the years, distillation has remained one of the most critical separation technologies used in various industries. Despite being one of the oldest separation methods, it is still relied upon where high purity and large volumes are required. Only a few technologies can match its flexibility and reliability at scale.
However, there is a clear difference between understanding the concept of distillation and operating it in real industrial conditions. At scale, the distillation process must handle continuous operation, high energy demand, and complex equipment behavior. The truth is, what works in a laboratory does not automatically work in an industrial plant.

Industries such as chemical, petrochemical, pharmaceutical, and energy processing operate under constant pressure to reduce energy consumption, control operating costs, and maintain product quality. Separation failures can lead to significant losses, off-spec products, and increased regulatory risk. This makes reliable separation technologies non-negotiable in modern plants.
Under these conditions, distillation often becomes the only viable solution capable of delivering high purity and stable performance at industrial scale. This guide examines what distillation is, why it remains essential despite its energy intensity. Also, it addresses how the distillation process evolves from a basic scientific principle into a large-scale industrial operation.
What Is Distillation?
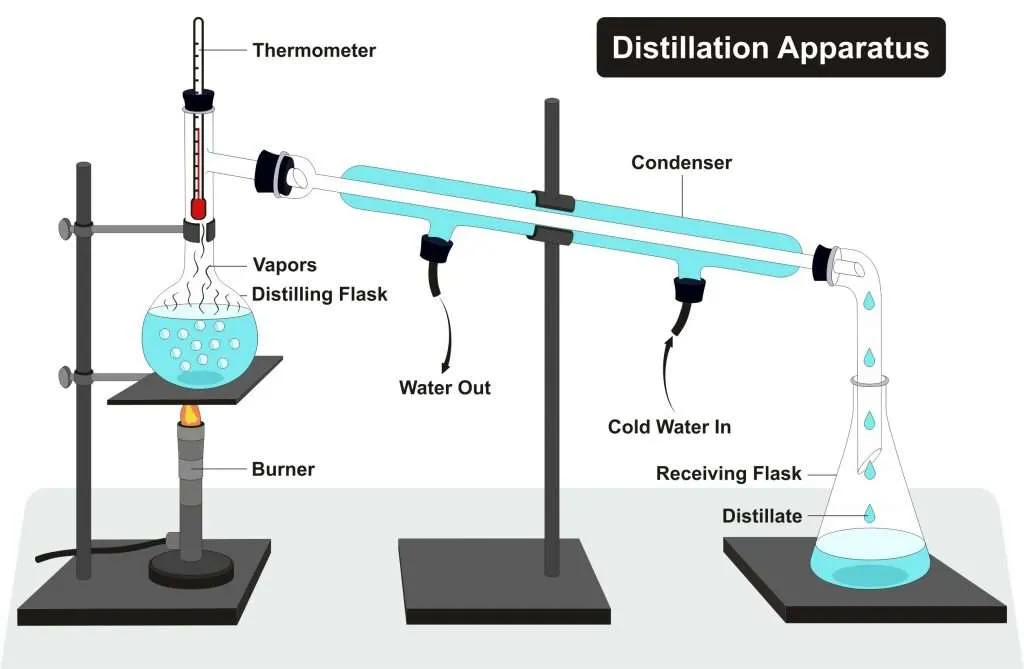
Distillation is a separation process driven by heat. It works by exploiting differences in boiling points within a liquid mixture. When heat is applied, components vaporize at different rates, allowing them to be separated through controlled evaporation and condensation.
This simple principle forms the basis of the distillation process used in both laboratory and industrial distillation systems.
What Is Industrial Distillation?
Industrial distillation is the application of the distillation process on a large scale. It uses engineered distillation equipment designed to separate liquid mixtures with different boiling points under controlled industrial conditions.
Unlike laboratory distillation, industrial distillation operates at high flow rates. It handles large volumes over long periods. Energy input, pressure, and safety are tightly controlled. The goal is stable and repeatable production.
The fundamental difference lies in heat recovery and continuity. Industrial systems are built to recover and reuse energy. They are also designed for continuous operation. This improves efficiency and reduces operating cost.
Because of its scale and complexity, industrial distillation is a core unit operation. It is essential in chemical, petrochemical, pharmaceutical, and energy industries. Separation performance directly affects cost, efficiency, and plant reliability.
The Industrial Distillation Process

Below is a step-by-step process of distillation:
Feed Preparation Under Industrial Conditions
In industrial distillation, the feed is rarely a clean or simple stream. It often contains a liquid mixture made up of two or more liquids, dissolved solids, or trace impurities. Before entering the distillation column, the feed is conditioned to ensure stable operation. This may include filtration, phase adjustment, or ratio control to maintain a consistent liquid ratio and protect downstream distillation equipment.
Heat Input and Energy Recovery
The distillation process begins with controlled heat input. Distillation involves boiling, but at an industrial level, heat transfer must be efficient and predictable. Heat is added until the boiling liquid reaches its required boiling temperature, which depends on vapor pressure, atmospheric pressure or reduced pressure conditions. To reduce energy use, large-scale systems rely heavily on heat recovery, reboilers, and integration between hot and cold streams.
Vapor–Liquid Mass Transfer
As the heated mixture enters the column, a vapor mixture forms. Rising vapour moves upward while liquid flows downward, creating continuous contact between phases. Separation occurs because of the difference in boiling points and partial pressure behavior of the components. The more volatile component concentrates in the vapor phase, while less volatile material remains in the remaining liquid. This vapor–liquid interaction is central to separation efficiency.
Product Withdrawal and Purity Control
Separated components leave the system as liquid fractions after condensation. The condensed liquidis collected as a purified liquid or sent for further distillation if higher purity is required. Careful control of withdrawal points ensures stable quality across industrial applications, whether the goal is solvent recovery, fuel production, or chemical purification.
Continuous Operation Stability
Most large scale industrial distillation systems operate under continuous distillation mode. Stability is critical. Flow rates, temperature profiles, and pressure must remain steady to avoid off-spec products. Unlike laboratory setups using a Bunsen burner flame or simple cooling with cold water, industrial systems depend on automated control to maintain long-term reliability in large scale industrial environments.
Key Types of Industrial Distillation
Industrial distillation is applied in different forms depending on the nature of the mixture, operating conditions, and desired product purity. Several types of distillation are applied depending on separation needs. Below are the most common types of distillation:
Simple Distillation
Simple distillation is used when the difference in boiling points between components is sufficiently large. Under such conditions, separation can be achieved with minimal equilibrium stages.
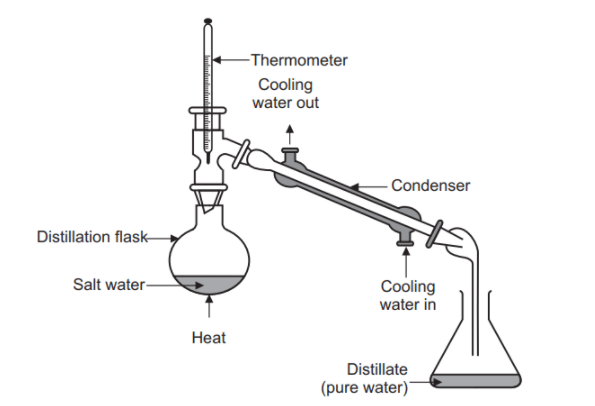
Its industrial relevance is limited. Simple distillation is mostly applied in small-scale operations, laboratory environments, or as a pre-separation step before more advanced distillation processes. It is rarely used as a standalone method in large industrial systems.
Fractional Distillation
Fractional distillation is the dominant method of industrial distillation. It is designed to separate mixtures with relatively close boiling points by creating multiple stages of vapor–liquid equilibrium.

This process is carried out in distillation columns equipped with trays or structured packing, which enhance contact between rising vapor and descending liquid. Separation efficiency improves across these repeated equilibrium stages.
Fractional distillation typically operates continuously at scale, making it essential in industries such as petroleum refining (where crude oil is separated into usable products), petrochemicals, and chemical manufacturing. In essence, it is used when boiling points are relatively close.
Vacuum Distillation
Vacuum distillation is applied to heat-sensitive or high-boiling materials that would degrade under normal atmospheric conditions.
By operating at reduced pressure, the boiling temperatures of components are lowered. This allows separation to occur without exposing the mixture to excessive heat, thereby preventing thermal degradation and preserving product quality.
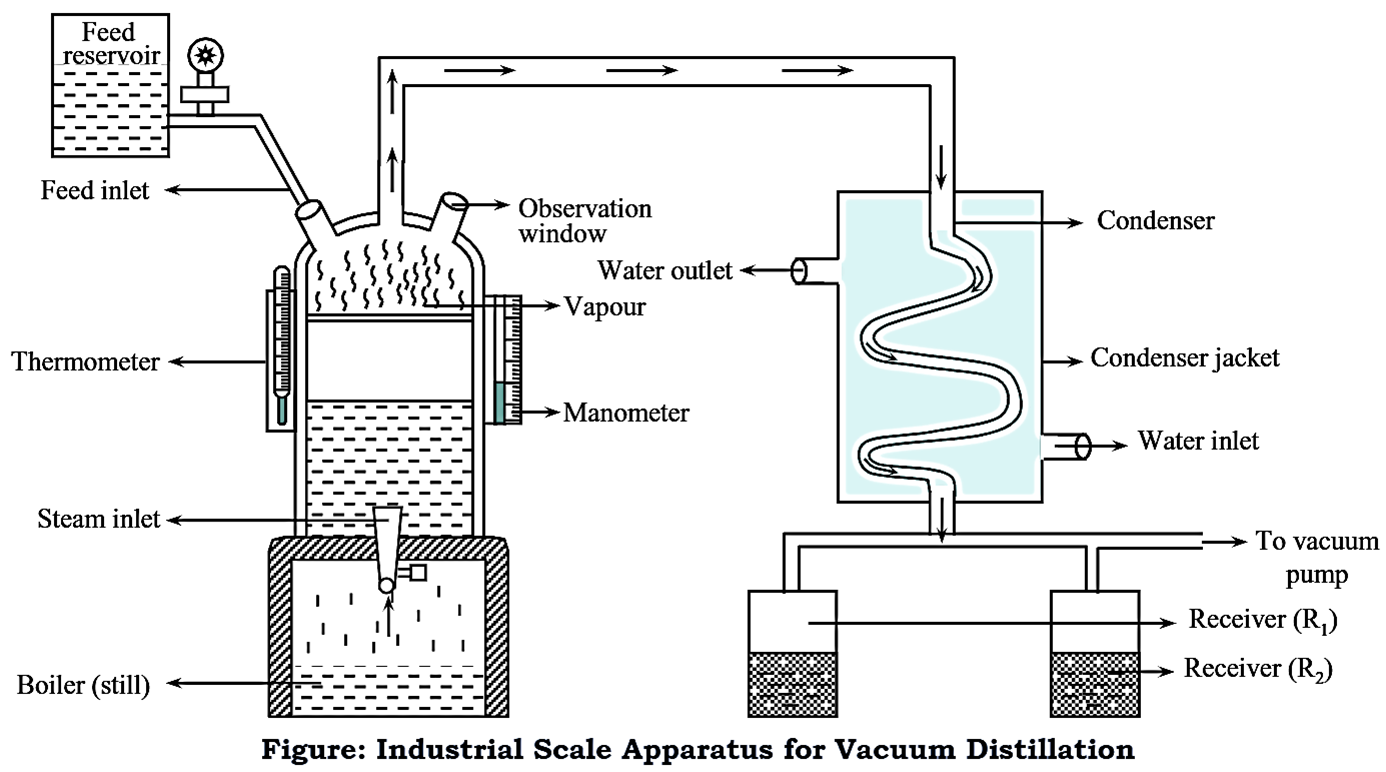
Vacuum distillation is especially important in heavy oil processing and specialty chemical production. Importantly, it is used in downstream processing of crude oil fractions.
Reactive Distillation
In reactive distillation, chemical reaction and separation occur simultaneously within the same unit. Reactants are converted into products while the products are continuously removed through distillation.
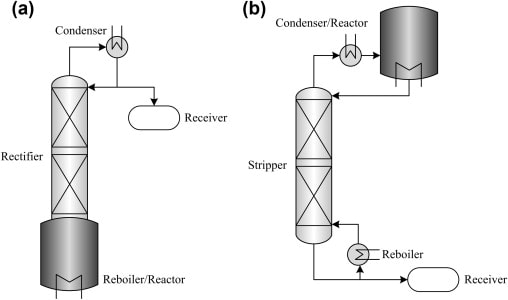
This approach is considered a process-intensification technique rather than a universal replacement for conventional distillation. While it can improve efficiency and reduce equipment size, its application is limited to systems where reaction and separation conditions are compatible.
Challenges in Traditional Industrial Distillation
1: Energy Consumption
Distillation is an energy-intensive process, often accounting for 30–40% of the total energy consumption in chemical plants. Large amounts of heat are required to vaporize liquid mixtures and maintain steady operation across distillation columns.
As a result, energy cost frequently represents the largest operating expense in industrial distillation systems. This has made energy efficiency a central concern, especially in industries facing rising fuel prices and stricter sustainability targets.
2: Space & Capital Intensity
Traditional distillation systems require tall distillation towers to achieve sufficient vapor–liquid contact and separation efficiency. These large structures demand substantial physical space and robust mechanical support.
Operational constraints such as flooding limitations further restrict throughput and column performance. In addition, the need for heavy foundations, structural steel, and complex installation leads to high civil and installation costs, increasing the overall capital investment required for distillation units.
3: Product Degradation
Extended residence time within the column and exposure to high operating temperatures can negatively affect sensitive products. Thermal stress may lead to decomposition, loss of purity, or reduced functional performance.
This challenge is particularly critical in industries such as pharmaceutical manufacturing and battery electrolyte production, where product stability and chemical integrity are essential. In such cases, traditional distillation may require modification or replacement with alternative separation technologies.
Industrial Distillation Applications by Industry
Solvent Recovery Distillation in Chemical and Battery Industries
Solvent recovery is one of the most important industrial distillation process applications today. It focuses on separating and purifying solvents based on differences in boiling points.
In this distillation process, contaminated solvents are heated and separated for reuse. Fractional distillation is commonly applied when the liquid mixture contains multiple components with close boiling points.
This industrial distillation process reduces raw material costs and minimizes waste. It also supports sustainability goals and regulatory compliance.
Stable operation, high purity, and energy efficiency are essential. These requirements make fractional distillation the preferred distillation process in chemical and battery industries.
Extractive Distillation for Difficult Separations
Extractive distillation is applied when differences in boiling points are too small for standard separation. Conventional fractional distillation becomes ineffective under these conditions.
In this specialized distillation process, a solvent is added to modify relative volatility. This allows separation of azeotropes and close-boiling systems.
The method extends the limits of fractional distillation. It is used selectively due to higher complexity and operating cost.
The Future of Industrial Distillation
HiGee (High-Gravity) Distillation
Limitations of Traditional Distillation
Traditional industrial distillation is largely gravity-driven. Conventional systems such as fractional distillation columns rely on height, residence time, and repeated vapor–liquid equilibrium stages to separate liquid mixtures based on differences in boiling points. This is increasingly constrained by energy consumption, equipment size, and thermal stress on products.
As operating pressures rise and separation tasks involve close boiling points, very high boiling points, or heat-sensitive materials, gravity-based distillation reaches practical limits. In many cases, vacuum operation becomes necessary, increasing reliance on vacuum pumps and adding system complexity, parasitic energy consumption, and maintenance burden. Tall distillation towers, long vapor paths, and extended exposure to high temperatures reduce separation efficiency and increase operating cost. These limitations have driven the need for advanced distillation processes that go beyond traditional gravity dependence.
It is within this context that HiGee (High-Gravity) Heat Pump Distillation was developed—integrating process intensification with energy recovery to overcome the structural and thermodynamic limits of conventional distillation.
What Is VHPS (HiGee Heat Pump Distillation)?

HiGee Heat Pump Distillation, also referred to as VHPS, combines High-Gravity (HiGee) process intensification with heat pump technology to redefine how the distillation process is performed at industrial scale.
High-Gravity (HiGee) Process Intensification
HiGee technology replaces gravity with strong centrifugal force to drive vapor–liquid contact. This dramatically increases mass transfer rates compared to traditional fractional distillation. Separation occurs over much shorter distances, even for liquid mixtures with small differences in boiling points. As a result, residence time is reduced and separation efficiency is improved within a compact system.
Mechanical Vapor Recompression (MVR) Heat Pump Integration
The heat pump component captures and reuses latent heat from the vapor phase. Instead of rejecting energy, the system recompresses vapor and feeds it back into the distillation process. This reduces external heat demand and stabilizes boiling temperature control, making the distillation process far more energy efficient than conventional systems.
Addressing Core Distillation Challenges
- Energy Consumption
In traditional distillation, energy demand rises sharply when separating mixtures with close boiling points. HiGee Heat Pump Distillation minimizes this penalty by combining intensified mass transfer with heat recovery. The result is a significant reduction in total energy consumption compared to conventional fractional distillation systems. - Equipment Footprint
Gravity-based distillation requires tall columns to achieve separation. HiGee systems achieve the samE performance in much smaller units. The reduced footprint lowers capital cost, simplifies installation, and makes large-scale industrial distillation feasible even in space-constrained facilities. - Product Degradation
Extended residence time and high temperatures often lead to degradation in traditional distillation, especially for pharmaceuticals, battery electrolytes, and specialty chemicals. HiGee Heat Pump Distillation shortens exposure time and allows separation at lower effective boiling temperatures, preserving product quality while maintaining separation efficiency.
Why This Represents the Next Stage of Industrial Distillation
By addressing energy intensity, equipment size, and thermal degradation simultaneously, HiGee Heat Pump Distillation represents a fundamental shift in how industrial distillation systems are designed. Rather than scaling up gravity-based columns, it re-engineers the distillation process itself, making high-performance separation possible with less energy, less space, and greater control.
Conclusion
Industrial distillation remains one of the most critical separation technologies in modern industry. At the same time, it is one of the most energy-intensive unit operations. The distillation process, driven by differences in boiling points, continues to underpin large-scale separation across multiple sectors.
Solvent recovery is now the dominant industrial application of distillation. Rising energy costs, stricter regulations, and sustainability goals have pushed fractional distillation systems toward higher efficiency, tighter control, and continuous operation. Reliable purification of liquid mixtures remains essential for industrial productivity.
Looking ahead, energy efficiency and process intensification are reshaping system design. Advances in equipment, control strategies, and alternative distillation configurations aim to reduce energy demand while preserving separation performance. The future of industrial distillation lies in doing more with less, without compromising purity or reliability.
Ready to Explore the Future of Industrial Distillation?
Are you a student, a process engineer or a technical manager who wants to understand the industrial distillation process and it efficiency? You’re in the right place. Here are everything you need to know about distillation and other separation techniques.
Beyond that, Jiangsu Super Distillation Technology Co., Ltd. delivers advanced HiGee Heat-Pump Distillation systems designed for high-efficiency solvent recovery, purification, and process intensification at an industrial scale.
Discover how energy-efficient, compact distillation technologies are redefining modern separation by paying a visit to our website and contacting us today to explore expert articles, case studies, and practical solutions designed for professionals like you.





